Researchers at the Institute of Automation of the Chinese Academy of Sciences have developed a compact, battery-powered brain stimulation device capable of delivering therapeutic magnetic pulses while a person is walking or performing everyday activities, according to Medical Xpress.
Repetitive transcranial magnetic stimulation is used to treat conditions such as depression, stroke-related motor impairment, and other neuropsychiatric disorders. It is also used in cognitive and motor function research.
Existing systems need to be plugged into a power supply and have bulky designs meant for stationary use in clinical settings. These limitations prevent stimulation during natural movement, such as standing and walking, making at-home or on-the-go treatments impractical.
Investigations into how brain activity changes in dynamic environments remain incomplete because current devices cannot be used during free behaviors. Patients undergoing treatment must attend daily hospital visits over extended periods, which affects adherence and increases cost. No battery-powered or wearable system has previously achieved the intensity levels needed to trigger direct neuronal responses in the brain.
In the study, "A wearable repetitive transcranial magnetic stimulation device," published in Nature Communications, researchers developed and tested a portable brain stimulation system by combining lightweight coil engineering with high-voltage pulse technology to enable therapeutic stimulation during natural human movement.
A total of 15 healthy adults between the ages of 25 and 40 participated in a human testing phase.
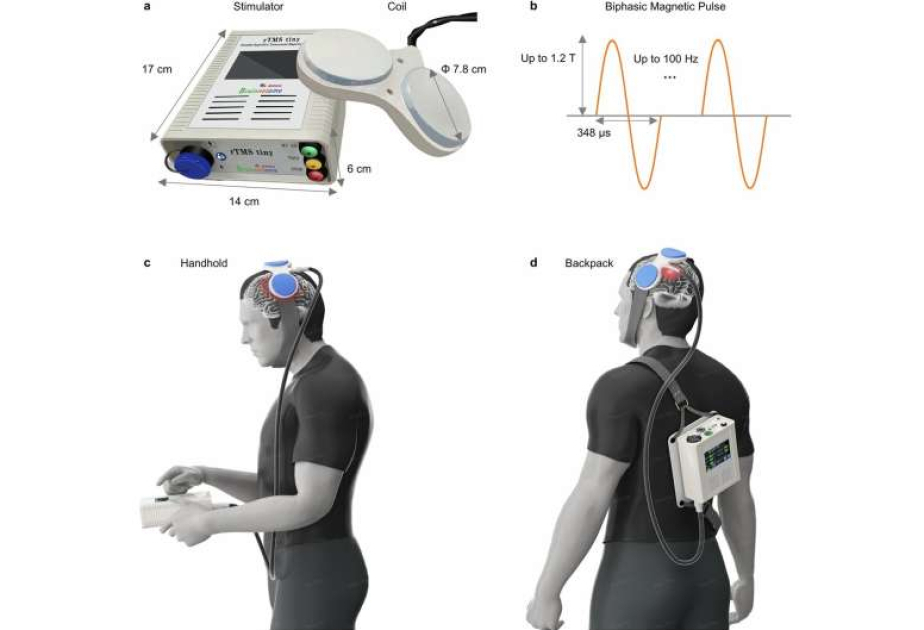
Researchers began by designing a head-mounted coil capable of producing strong magnetic fields while using only battery power. Traditional systems rely on high-voltage electricity from the grid, generate high heat, and weigh too much for wearable applications. To address this challenge, the team engineered a novel compact figure-8 coil using specialized magnetic core materials that enhance the magnetic field while reducing energy requirements.
A control and power unit, weighing just over a kilogram, was secured across the back using shoulder straps. A high-power converter was built in to deliver pulses of up to 1,600 volts and 500 milliamps using an 18-volt battery. The unit recovered energy after each pulse to extend battery life and limit heat generation.
The coil was mounted onto the heads of participants using a padded elastic strap that wrapped under the chin for stability. This made the entire system wearable without restricting normal standing or walking movements.
Tests included simulations of electric field strength in human head models and comparisons with a commercial device. Pulse width, magnetic flux intensity, temperature rise, and repetition rates were all measured and compared to conventional systems.
Human testing followed the technical validation with participants receiving magnetic stimulation to their motor cortex while seated and while walking. To verify that the brain was responding, motor-evoked potentials were recorded from muscles in the hands and legs to confirm that the magnetic pulses reached motor areas of the brain.
In walking conditions, muscle responses more than doubled in amplitude compared to those measured at rest. These results confirmed that the device delivered effective stimulation even during natural movement.
Magnetic pulses generated by the wearable coil reached 1.2 Tesla with a pulse width of 348 microseconds, comparable to commercial brain stimulation systems. The system operated at frequencies up to 100 Hz and supported standard stimulation protocols including 10 Hz and theta burst patterns.
Battery-powered operation allowed delivery of over 8,000 pulses at moderate intensity on a single charge. Coil temperatures remained below safety thresholds defined by international medical device standards during high-frequency use. Infrared imaging showed uniform heat distribution, and no overheating occurred even after extended sessions.
While all testing was conducted in controlled laboratory settings, the authors propose that future versions of the device may support home-based or mobile use, pending further development of safety protocols and remote supervision systems.
Plans for future use include integration with brain-monitoring systems to allow real-time feedback and adaptive stimulation, which could support closed-loop therapeutic applications.